Research and Innovation Services Partner with us Our Technologies Wound Healing Peptide
Wound Healing Peptide
- Aboriginal and Torres Strait Islander in Marine Science
- Courses
- Future Students
- Current Students
- Research and Teaching
- Partners and Community
- About JCU
- Reputation and Experience
- Celebrating 50 Years
- Academy
- Anthropological Laboratory for Tropical Audiovisual Research (ALTAR)
- Anton Breinl Research Centre
- Agriculture Technology and Adoption Centre (AgTAC)
- Living on Campus
- How to apply
- Advanced Analytical Centre
- Alumni
- AMHHEC
- Aquaculture Solutions
- AusAsian Mental Health Research Group
- ARCSTA
- Area 61
- Association of Australian University Secretaries
- Australian Lions Stinger Research
- Australian Tropical Herbarium
- Australian Quantum & Classical Transport Physics Group
- Boating and Diving
- JCU-CSIRO Partnership
- Employability Edge
- Clinical Psychedelic Research Lab
- Centre for Tropical Biosecurity
- Career Ready Plan
- Careers at JCU
- Careers and Employability
- Chancellery
- Centre for Tropical Bioinformatics and Molecular Biology
- CITBA
- CMT
- CASE
- College of Business, Law and Governance
- College of Healthcare Sciences
- College of Medicine and Dentistry
- College of Science and Engineering
- CPHMVS
- Centre for Disaster Solutions
- CSTFA
- Cyber Security Hub
- Cyclone Testing Station
- The Centre for Disaster Studies
- Daintree Rainforest Observatory
- Defence
- Discover Nature at JCU
- Research Division
- Services and Resources Division
- Education Division
- Elite Athletes
- eResearch
- Environmental Research Complex [ERC]
- Estate
- Fletcherview
- Foundation for Australian Literary Studies
- Gender Equity Action and Research
- General Practice and Rural Medicine
- JCU Orientation
- Give to JCU
- Governance
- Art of Academic Writing
- Art of Academic Editing
- Graduate Research School
- Graduation
- Indigenous Education and Research Centre
- Indigenous Engagement
- Indigenous Legal Needs Project
- Inherent Requirements
- IsoTropics Geochemistry Lab
- IT Services
- International Students
-
Research and Innovation Services
- About Research and Innovation Services
- Partner with us
- Innovate with us
- Research Grants, Tenders and Funding
- Ethics and Research Integrity
- Research Contracts
- Discover our Research and Testing Facilities
- JCU Ideas Lab
- Find an Expert
- Contact Research and Innovation Services
- FAQs and Fact Sheets
- GECO
- JCU Eduquarium
- JCU Heroes Programs
- JCU Webinars
- JCU Events
- JCU Global Experience
- JCU Ideas Lab
- JCU Job Ready
- JCU Motorsports
- JCU Prizes
- JCU Sport
- JCU Turtle Health Research
- Language and Culture Research Centre
- CEE
- LearnJCU
- Library
- Mabo Decision: 30 years on
- MARF
- Marine Geophysics Laboratory
- New students
- Off-Campus Students
- Office of the Vice Chancellor and President
- Virtual Open Day
- Orpheus
- Open Day
- Outstanding Alumni
- Parents and Partners
- Pathways to university
- Pharmacy Full Scope
- Planning for your future
- Placements
- Policy
- PAHL
- Publications
- Professional Experience Placement
- Queensland Research Centre for Peripheral Vascular Disease
- Rapid Assessment Unit
- RDIM
- Researcher Development Portal
- Roderick Centre for Australian Literature and Creative Writing
- Safety and Wellbeing
- Scholarships
- Contextual Science for Tropical Coastal Ecosystems
- Staff
- State of the Tropics
- Strategic Procurement
- Student Equity and Wellbeing
- Student profiles
- SWIRLnet
- TARL
- TESS
- TREAD
- TropEco for Staff and Students
- TQ Maths Hub
- TUDLab
- Unicare Centre and Unicampus Kids
- UAV
- VAVS Home
- Work Health and Safety
- WHOCC for Vector-borne & NTDs
- Media
- Copyright and Terms of Use
- Australian Institute of Tropical Health & Medicine
- Pay review
Background
Poor wound healing after trauma, surgery, acute illness, or chronic disease conditions affects millions of people worldwide each year.
In particular, foot ulcers are a common complication of poorly controlled diabetes. Diabetic foot ulcers (DFUs) respond poorly to treatment and often require limb amputation as a life-saving procedure, adding clinical and psychological consequences of limb loss to morbidity caused by ulcers.
Despite a large variety of wound care products, only 3 products have been approved by the Food and Drug Administration (FDA) for the treatment of diabetic foot ulcers. Of these, 2 are bioengineered skin-cares products, and the third is a growth-factor whose healing rates are 36-50% after 20 weeks. It is critical to develop a better wound dressing material that can promote local tissue regeneration.
The underlying discovery came from study of Opisthorchiasis, which is a tropical disease caused by ingestion of the food-borne parasite Opisthorchis viverrini (also known as the Southeast Asian liver fluke). JCU researchers found the parasite secretes a peptide, named Ov-GRN-1, to repair damage it causes in bile ducts from feeding. Ov-GRN-1 belongs to a secreted growth factor family found in a majority of organisms. Granulin-like proteins are heavily involved in wound healing. The original protein has complex knotted protein structure. JCU researchers generated sets of synthesised peptides derived from Ov‑GRN-1 and screened them for increased stability, improved production yield, and enhanced activity in vitro and in vivo. Of these, JCU303, a minimal 24-mer peptide was found to have superior wound healing properties, more stability and reduced immunogenic potential compared to the parent protein.
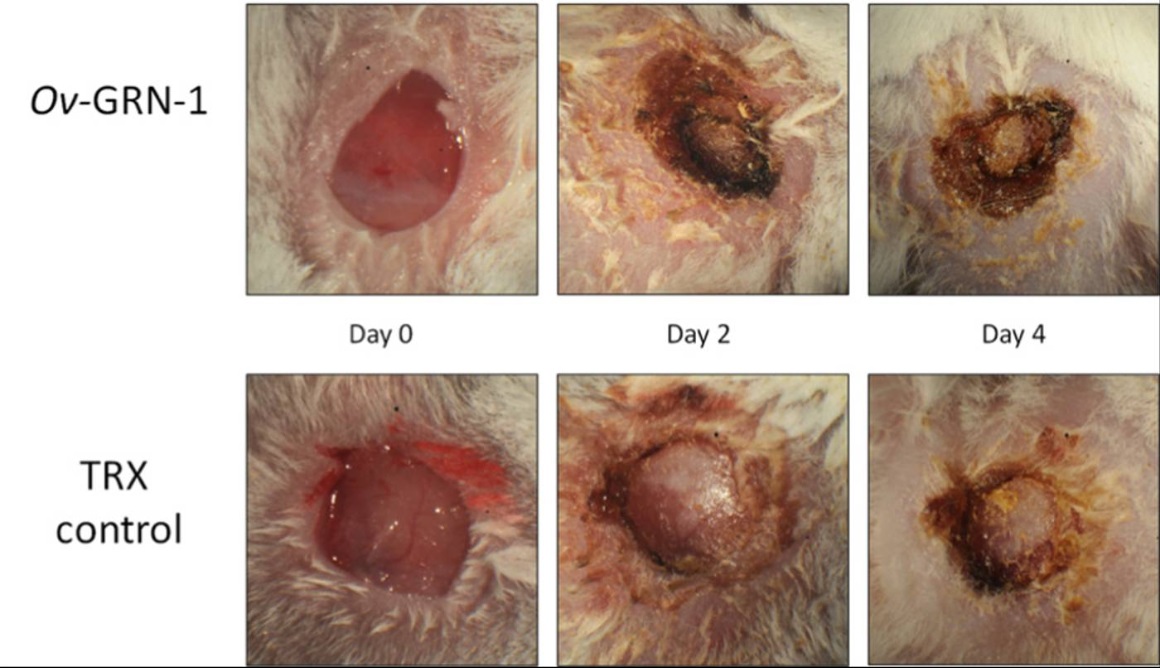
Figure 1.
- Efficacy: 20% faster healing than nearest competitor in animal studies
- Safety: May offer safety advantages
- Indication: Potentially broader indications
- Cost: Peptide manufacturing easier & less expensive than biologics
- Activity
- Stimulates fibroblast migration & proliferation, collagen production
- Accelerates wound re-epithelialisation
- Restores depleted growth factor activity
- Promotes re-vascularisation
Topical treatment of:
- Chronic wounds
- Diabetic foot ulcer
- Pressure Ulcers
- Venous Stasis Ulcers
- Burns
- Surgical wounds
We are seeking partners for continued development of the lead candidate through preclinical development & clinical trials.
Seeking:
- Development partners
- Commercial partner
- Licensing
- University spin out
- Seeking investment
IP Status
- Patent application submitted
Patent
- PCT/AU2017/050959